 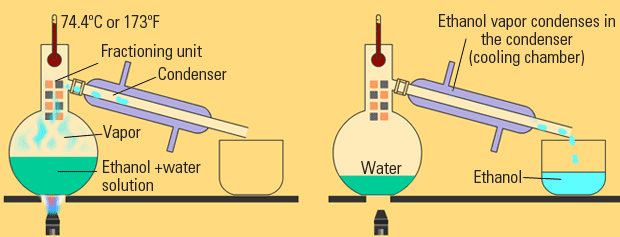
Since distillation depends on the different vapor pressures of the components to be separated, let's first consider the. Fractional distillation is used to purify chemicals and to separate mixtures to obtain their components. If the mole fraction of A in the mixture is A, then by the definition of mole fraction, that of B is. Fractional distillation is a process by which components in a chemical mixture are separated into different parts (called fractions) according to their different boiling points. In the present paper, a membrane distillation and crystallization (MDCr) process was proposed to achieve zero liquid discharge (ZLD) of aniline wastewater. To fully understand distillation, we will consider an ideal binary liquid mixture of A and B. It is then cooled to 200 degrees where it becomes a liquid, therefore we can say it has been liquefied. Aniline is a highly toxic organic pollutant with carcinogenic, teratogenic and mutagenesis characteristics. This is the first step in the processing of crude oil, and it is considered to be the main separation process as it performs the initial rough. Before entering the fractionating column, air is filtered to remove any excess particles such as dust. Fractional distillation is the process by which oil refineries separate crude oil into different, more useful hydrocarbon products based on their relative molecular weights in a distillation tower. Fractional distillation is a process by which components in a chemical mixture are separated into different parts (called fractions) according to their different boiling points. Nitrogen and oxygen in air can be separated by the process of fractional distillation. 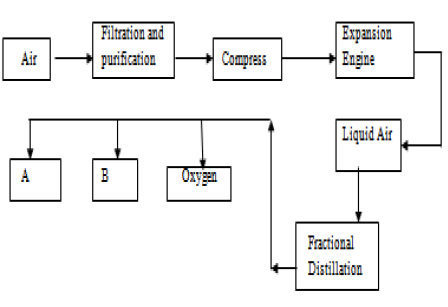
Oxygen makes up one-fifth of the Earth’s atmosphere and the remaining 1% are made up of traces of carbon dioxide (0.04%), water vapour and noble gases, like argon (0.9%) It makes up about four-fifths of the Earth’s atmosphere. In Earth’s atmosphere, nitrogen is the most abundant gas. These amounts have not changed over the last 200 million years. The different gases in the atmosphere are found in different amounts. Gases are found in different proportions.In the Earth’s atmosphere today, there are a mixture of gases, including about 80% nitrogen, about 20% oxygen and various other gases. The atmosphere contains a mixture of gases.You are free to add your own answers below this page to assist others improve their learning.Gases in the Atmosphere (GCSE Chemistry) Atmospheric Gases Proportions of Atmospheric Gases Nitrogen is gotten in large quantity from fractional distillation of liquid air.Gaseous mixtures like air are firstly liquified before distilling fractionally.Fractional distillation is usually done on liquids.Evaporation will rather be used to separate sulphur from carbon disulphide. this can be manipulated to form a boundary layer separable by separating funnel method. At each distillation the vapour is richer in nitrogen (the component. Evaporation will rather be appropriate here. The liquid air is separated into its constituent components by fractional distillation. Nitrogen is obtained from liquid air by fractional distillation. Now for the right answer to the above question: RELATED => The process whereby some organisms with certain. Air is firstly liquified by subjecting to intense pressure and reduced temperature, and then the resultant liquified air is subjected to fractional distillation to separate it into its components which are: Nitrogen, oxygen, carbon dioxide, and noble gasses. The key word here is liquids of different boiling points. Filtration is a method for separating an insoluble solid from a liquid. For example, sand does not dissolve in water it is insoluble. Heat is usually applied to take advantage of the differences in boiling points of each of the components to isolate them beginning from the component with the lowest boiling point. SEPARATION TECHNIQUES If a substance does not dissolve in a solvent, we say that it is insoluble. Sulphur from the solution of sulphur in carbon disulphide QUICK ANSWER…įractional distillation is a separation technique used to separate two or more liquids of different boiling points. Iodine from solution of Iodine in carbon tetrachlorideĭ. Which of the following can be obtained by fractional distillation?Ĭ. In the diagram above, a bulb is lit by drawing 2.0A from 440V a.c.Find the simple interest on N225 in 5 years at 10%….The liquid air gets separated depending on the boiling point of the gas. The separated gases are obtained by the receiver ends from the fractional distillation column. Which of the following substances has the lowest vapor density? - (b) Since its a mixture of liquids, rather than a solid dissolved in a liquid, fractional distillation is used. Solution Verified by Toppr Correct option is B) The components of liquid air on passing through a fractional distillation column separate based on boiling point. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
